Key Points
- This multicenter clinical trial is evaluating the safety of using focused ultrasound to temporarily and reversibly open the blood-brain barrier (BBB) in glioblastoma patients undergoing standard chemotherapy treatment.
- Researchers hope that disrupting the BBB will enhance the delivery of chemotherapy to the tumor site.
- The study at the University of Virginia is being led by Jason Sheehan, MD, PhD, Professor of Neurological Surgery and Neuroscience and the Foundation’s Senior Advisor for Brain Tumor Research.
- The Foundation is contributing to this clinical trial through an ongoing partnership with the UVA Focused Ultrasound Center of Excellence.
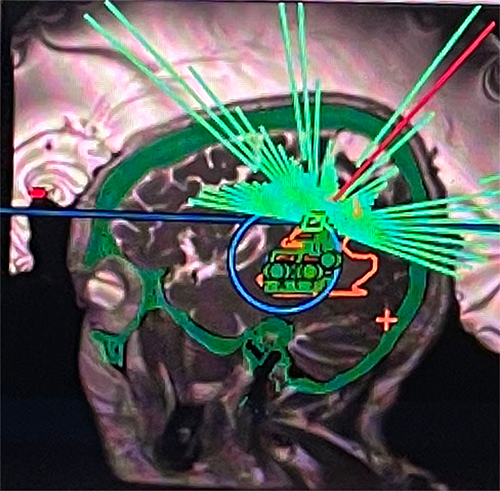 On June 22, 2021, a team at the University of Virginia (UVA Health) treated the first patient as part of a multicenter clinical trial evaluating the safety of using focused ultrasound to enhance the delivery of chemotherapy in patients with glioblastoma (GBM).
On June 22, 2021, a team at the University of Virginia (UVA Health) treated the first patient as part of a multicenter clinical trial evaluating the safety of using focused ultrasound to enhance the delivery of chemotherapy in patients with glioblastoma (GBM).
The study at UVA Health is being led by Jason Sheehan, MD, PhD, Professor of Neurological Surgery and Neuroscience. Dr. Sheehan also serves as the Foundation’s Senior Advisor for Brain Tumor Research.
GBMs are aggressive, malignant tumors that form in the brain and spinal cord. Traditional treatment involves surgery followed by chemotherapy and radiation. The median survival of patients after a GBM diagnosis is only 15-18 months.
One of the reasons traditional treatment is not more effective is because the brain has a protective layer of tightly joined cells called the blood-brain barrier (BBB) that normally prevents harmful substances, such as toxins and infectious agents, from diffusing from the blood into the surrounding brain tissue. However, the BBB can also prevent beneficial therapeutic agents, like chemotherapy, from getting into the brain.
The trial is investigating the use of low intensity focused ultrasound to temporarily and reversibly open the BBB in GBM patients undergoing standard chemotherapy treatment.
Researchers hope that opening the BBB will allow chemotherapy to enter the brain at the tumor site in higher concentrations than would normally occur.
“MRI-guided focused ultrasound represents an entirely new approach to treating patients with complex neuro-oncologic problems,” says Dr. Sheehan. “In this trial, low intensity focused ultrasound opens a transient window in the BBB that can augment delivery of chemotherapy precisely to the brain tumor. We look forward to treating additional patients as part of this and several other focused ultrasound brain tumor trials.”
The clinical trial uses Insightec’s investigational Exablate Neuro device, and – in addition to UVA – the study is ongoing at the University of Maryland, Brigham and Women’s Hospital, and West Virginia University.
The Foundation is contributing to this clinical trial through an ongoing partnership with the UVA Focused Ultrasound Center of Excellence.
“We were very impressed with the accuracy and precision of the BBB opening after focused ultrasound treatment,” said Dr. Sheehan.
“Using focused ultrasound to address glioblastoma is one of our top research priorities,” says Foundation Chairman Neal F. Kassell, MD. “This trial is one in our portfolio of ongoing preclinical laboratory studies and clinical trials investigating focused ultrasound for GBM. We also recently held our second workshop on the topic, convening critical stakeholders to share knowledge, identify gaps in evidence, and create a roadmap necessary for focused ultrasound to progress in the treatment of GBM. We are encouraged by this first treatment at UVA, and we applaud the pioneering work of researchers at all of the participating sites.”
For Patients
This trial is actively recruiting patients. Those who are interested in learning more about this trial are encouraged to contact the sites below:
University of Virginia | Charlottesville, VA
Matthew Patterson
434-243-7336
mwp5f@hscmail.mcc.virginia.edu
Brigham and Women’s Hospital | Boston, MA
Anna Ball
617-732-6992
aball@bwh.harvard.edu
Ana Montalvo Landivar
754-422-3432
amontalvolandivar@bwh.harvard.edu
University of Maryland | Baltimore, MD
Kaitlyn Henry
410-328-0939
KHenry@som.umaryland.edu
Charlie Klontz
CMKlontz@som.umaryland.edu
West Virginia University | Morgantown, WV
Sylvia
304-293-1683
smcewuen@hsc.wvu.edu
