Key Points
- A pediatric patient participated in a focused ultrasound clinical trial for a disabling, painful, benign bone tumor.
- Now, five years post-treatment, she remains free from pain.
- In November of 2020, focused ultrasound earned FDA HDE approval to treat osteoid osteoma.
The Patient
 A 10-year-old female with no significant past medical or surgical history presented with a 7-month history of left hip and leg pain. There was no inciting injury, and the initial leg and hip X-rays were normal. The patient described the pain to be worse at night, often preventing her from sleeping and sometimes waking her up. She used to be a very active young girl, competing on a swim team year round, but she had to quit because of her pain. Her school attendance was also suffering due to pain and lack of sleep.
A 10-year-old female with no significant past medical or surgical history presented with a 7-month history of left hip and leg pain. There was no inciting injury, and the initial leg and hip X-rays were normal. The patient described the pain to be worse at night, often preventing her from sleeping and sometimes waking her up. She used to be a very active young girl, competing on a swim team year round, but she had to quit because of her pain. Her school attendance was also suffering due to pain and lack of sleep.
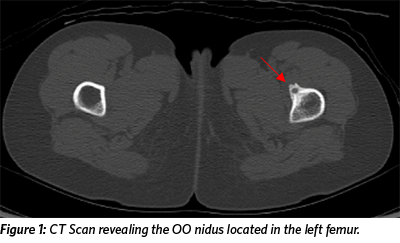
The patient reported taking Ibuprofen (400mg every eight hours) for pain control. A CT scan revealed a small, round, well-defined bone lesion in the proximal left femur, consistent with osteoid osteoma (OO) (Figure 1). MRI with contrast (not shown) showed enhancement within the nidus and edema in the adjacent bone marrow and soft tissues, also consistent with OO.
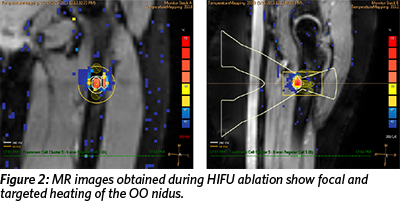
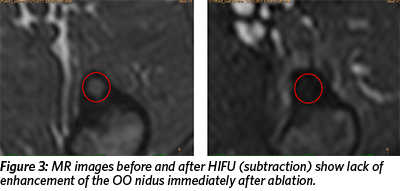
The patient enrolled in the Focused Ultrasound Osteoid Osteoma clinical trial at Children’s National Medical Center in Washington, DC. She underwent a one-time procedure of MR-guided high intensity focused ultrasound (HIFU) ablation of the OO under sedation. MR images obtained during the treatment revealed focal and targeted heating of the OO nidus (Figure 2) and lack of enhancement of the OO nidus immediately after ablation, confirming successful treatment (Figure 3).
 On the day after the treatment, the patient reported local pain and soreness that was relieved with Ibuprofen. Her pain gradually improved over the next week, and by the seventh day post-HIFU, her pain was completely gone, no longer requiring any medication. She was able to rejoin her swim team and return to school. The one-year follow-up MRI was normal with complete healing of the OO nidus. At five years post treatment, she remains completely cured. Per the patient’s mother, “She’s a normal teenager now.”
On the day after the treatment, the patient reported local pain and soreness that was relieved with Ibuprofen. Her pain gradually improved over the next week, and by the seventh day post-HIFU, her pain was completely gone, no longer requiring any medication. She was able to rejoin her swim team and return to school. The one-year follow-up MRI was normal with complete healing of the OO nidus. At five years post treatment, she remains completely cured. Per the patient’s mother, “She’s a normal teenager now.”
What is Osteoid Osteoma?
OO is a benign but painful bone tumor that commonly occurs in the long bones of children and adolescents. OOs produce a substance called prostaglandin, which causes a local increase in blood flow, inflammation, and pain. The pain is worsened by irritation of adjacent normal bone tissue and is characteristically worse at night, often waking children from sleep. Treatment with nonsteroidal anti-inflammatory drugs (NSAIDs) as a type of pain medication relieves the discomfort associated with OO, but long-term use of NSAIDs can have harmful side effects. If untreated, some OOs can lead to disabling sequelae, such as growth disturbance, restricted joint motion, and bone deformity.
 Definitive treatment options for OO include surgical resection, radiofrequency ablation (RFA), cryoablation, and focused ultrasound ablation of the OO nidus. Surgical resection is an uncommon first line therapy due to difficulty identifying the lesions intraoperatively, which can lead to significant damage to surrounding healthy bone and soft tissue. CT-guided RFA is the current standard of care, where a needle is inserted into the OO nidus under CT guidance and then heated to 90 degrees Celsius to ablate the tissue. Although this method is successful (85 to 98 percent success rate), it is still minimally invasive with risk for complications and exposes the patient to ionizing radiation.
Definitive treatment options for OO include surgical resection, radiofrequency ablation (RFA), cryoablation, and focused ultrasound ablation of the OO nidus. Surgical resection is an uncommon first line therapy due to difficulty identifying the lesions intraoperatively, which can lead to significant damage to surrounding healthy bone and soft tissue. CT-guided RFA is the current standard of care, where a needle is inserted into the OO nidus under CT guidance and then heated to 90 degrees Celsius to ablate the tissue. Although this method is successful (85 to 98 percent success rate), it is still minimally invasive with risk for complications and exposes the patient to ionizing radiation.
Focused Ultrasound for Osteoid Osteoma
HIFU provides a precise, noninvasive, and radiation-free treatment for OO. Using MRI guidance and temperature mapping, the OO target can be clearly delineated and ablated without damage to surrounding normal tissue. There are numerous reports in the scientific literature that highlight HIFU as a safe and effective therapy for OO in both children and adults.
In November 2020, the Sonalleve device was approved under the US Food and Drug Administration’s Humanitarian Device Exemption (HDE) to treat osteoid osteoma in the extremities, marking the first focused ultrasound regulatory approval that directly impacts the pediatric population. Given its noninvasive, non-ionizing properties, HIFU could prove beneficial for children with various other medical conditions.
Recent Notable Research Results
A 2017 pilot study led by Dr. Sharma treated nine pediatric osteoid osteoma patients with MRI-guided HIFU and compared the outcomes to nine historical cases of OO treatment by RFA. At four weeks post-treatment, eight of nine participants that received MR-HIFU had complete resolution of OO-related pain and had stopped taking pain medication. There were no serious adverse events. The results showed that MR-HIFU ablation of painful OO is safe and feasible with clinical response rates similar to RFA.
In 2019, Arrigoni et al. reported the results of 33 children with OO treated with MRI-guided HIFU across three institutions. Thirty-two of the children reported complete pain relief within one week after the procedure. The child that still had residual pain after one week received an additional HIFU treatment and subsequently had complete resolution of pain.
Although HIFU is an attractive treatment option for OO, there remain limitations to the use of this device. One such limitation is the depth of the OO nidus within the bone. In the study by Arrigoni et al., the optimal depth of the lesion is no more than six millimeters from the bone surface. However, this still encompasses a large portion of OO patients because almost 50 percent fall within this category.
References
Sharma KV, Yarmolenko PS, Celik H, Eranki A, et al. Comparison of Noninvasive High-Intensity Focused Ultrasound with Radiofrequency Ablation of Osteoid Osteoma. J Ped 2017;190:222-228.
Arrigoni F, Napoli A, Bazzocchi A, Zugaro L, et al. Magnetic-resonance-guided focused ultrasound treatment of non-spinal osteoid osteoma in children: Multicentre experience. Ped Radiol 2019;49:1209-1216.
The Foundation thanks Karun Sharma, MD, of Children’s National Medical Center in Washington, DC, for providing this case report.
