Patients with localized prostate cancer are receiving treatment in a new clinical study assessing the safety of a transurethral ultrasound ablation system developed by Toronto-based Profound Medical Inc. The system, which uses a specially designed wand to deliver thermal ablative therapy, operates under MR guidance. It works on a variety of MRI platforms, can be moved from scanner to scanner and treats the whole prostate gland in one session.
The multi-center study, entitled Transurethral Ultrasound Ablation (TULSA), is expected to enroll 30 patients and follow them for one year. Trial collaborators are London Health Sciences Centre, Western University in Ontario and the Lawson Health Research Institute. Joseph L. Chin, MD, Professor of Urology and Oncology at Western University, is the Principal Investigator.
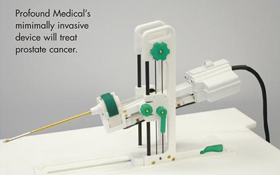 PMI CEO Steven Plymale explained, “The ive of our trial is to demonstrate that it’s possible to safely and effectively eliminate the cancerous tissue with decreased risk of side effects such as long-term impairment to bladder or bowel control, or erectile function.”
PMI CEO Steven Plymale explained, “The ive of our trial is to demonstrate that it’s possible to safely and effectively eliminate the cancerous tissue with decreased risk of side effects such as long-term impairment to bladder or bowel control, or erectile function.”
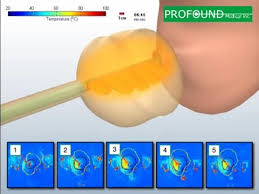 PMI’s technology is based on years of research and development, initially performed in collaboration with the Sunnybrook Research Institute in Toronto. To accelerate commercialization of its system, the company plans to collect clinical data in multiple jurisdictions at the same time. “The TULSA clinical trial marks a major milestone in our company’s evolution and the way that localized prostate cancer will be treated going forward,” said Plymale.
PMI’s technology is based on years of research and development, initially performed in collaboration with the Sunnybrook Research Institute in Toronto. To accelerate commercialization of its system, the company plans to collect clinical data in multiple jurisdictions at the same time. “The TULSA clinical trial marks a major milestone in our company’s evolution and the way that localized prostate cancer will be treated going forward,” said Plymale.
PROFOUND MEDICAL PRESS RELEASE
LONDON HEALTH SCIENCES CENTRE PRESS RELEASE AND VIDEO
Profound Medical, Inc. company profile (December, 2009, Focused Ultrasound Foundation Newsletter)
